Effect of lactobacillus in preventing post-antibiotic vulvovaginal candidiasis: a randomised controlled trial
BMJ 2004; 329 doi: https://doi.org/10.1136/bmj.38210.494977.DE (Published 02 September 2004) Cite this as: BMJ 2004;329:548- Marie Pirotta, senior lecturer (m.pirotta{at}unimelb.edu.au)1,
- Jane Gunn, associate professor1,
- Patty Chondros, statistician1,
- Sonia Grover, senior obstetrician2,
- Paula O'Malley, research assistant1,
- Susan Hurley, associate lecturer3,
- Suzanne Garland, professor, department of microbiology and infectious diseases2
- 1 Department of General Practice, 200 Berkeley Street, Carlton, Victoria, Australia, 3053
- 2 Royal Women's Hospital, Women's and Children's Health, 132 Grattan Street, Carlton, Victoria
- 3 School of Population Health, University of Melbourne, Swanston Street, Carlton, Victoria
- Correspondence to: M Pirotta
Abstract
Objective To test whether oral or vaginal lactobacillus can prevent vulvovaginitis after antibiotic treatment.
Design Randomised, placebo controlled, double blind, factorial 2×2 trial.
Setting Fifty general practices and 16 pharmacies in Melbourne, Australia.
Participants Non-pregnant women aged 18-50 years who required a short course of oral antibiotics for a non-gynaecological infection: 278 were enrolled in the study, and results were available for 235.
Interventions Lactobacillus preparations taken orally or vaginally, or both, from enrolment until four days after completion of their antibiotic course.
Main outcome measures Participants' reports of symptoms of post-antibiotic vulvovaginitis, with microbiological evidence of candidiasis provided by a self obtained vaginal swab.
Results Overall, 55/235 (23% (95% confidence interval 18% to 29%)) women developed post-antibiotic vulvovaginitis. Compared with placebo, the odds ratio for developing post-antibiotic vulvovaginitis with oral lactobacillus was 1.06 (95% confidence interval 0.58 to 1.94) and with vaginal lactobacillus 1.38 (0.75 to 2.54). Compliance with antibiotics and interventions was high. The trial was terminated after the second interim analysis because of lack of effect of the interventions. Given the data at this time, the chances of detecting a significant reduction in vulvovaginitis with oral or vaginal lactobacillus treatment were less than 0.032 and 0.0006 respectively if the trial proceeded to full enrolment.
Conclusions The use of oral or vaginal forms of lactobacillus to prevent post-antibiotic vulvovaginitis is not supported by these results. Further research on this subject is unlikely to be fruitful, unless new understandings about the pathogenesis of post-antibiotic vulvovaginitis indicate a possible role for lactobacillus.
Introduction
The use of many complementary and alternative medicines remains controversial. In Australia and the United States, the estimated annual expenditure associated with use of such treatments is $A2.3bn (£0.9bn) and $27.1bn (£15bn) respectively, which is more than that spent on prescription drugs.1 2
Probiotics, microorganisms that have antagonist activity against pathogens in vivo, have long been promoted as health enhancing in general and specifically as useful for vaginal problems.3 Probiotics are commonly used4–6 and recommended7 for vulvovaginitis that develops after antibiotic treatment—a condition of concern to many women4 and usually caused by Candida albicans.8 Lactobacilli are a genus of bacteria, many species of which have been evaluated for their probiotic potential and may be found in yoghurt. Our previous study found that 40% of a sample of 751 women with a history of vulvovaginitis had used yoghurt or lactobacillus orally or vaginally to prevent post-antibiotic vulvovaginitis,4 but no published trials have tested the effectiveness of this treatment.
The aim of our present study was to test whether oral or vaginal lactobacillus can prevent post-antibiotic vulvovaginitis.
Participants and methods
Participants
Recruitment took place in Melbourne, Australia, from November 2001 until July 2003, through 50 general practices and 16 pharmacies.9 Eligible women were aged 18-50 years with non-gynaecological infections who started short term treatment with oral antibiotics within 48 hours of enrolment. Women were ineligible if they had vaginal symptoms, had used vaginal antifungal agents within the past fortnight or other antibiotics within the past month, or were pregnant or immunocompromised. We obtained informed written consent from all participants.
Design
We used a 2×2 factorial design to test (a) an oral powder, either Lactobac (containing Lactobacillus rhamnosus and Bifidobacterium longum) or placebo (malto-dextrin powder) and (b) a vaginal pessary, either Femilac (containing L rhamnosus, L delbrueckii, L acidophilus, and Streptococcus thermophilus) or placebo (maltodextrin). Active treatments and placebos were identical in appearance. Participants took half a teaspoon of powder twice daily 20 minutes before meals and one pessary at night for 10 days (during the six day antibiotic course and for four days after).
Independent examination of the treatments on three occasions (by the microbiology laboratory of Royal Women's Hospital, Melbourne) confirmed lactobacillus viability and accuracy of labelling of active and placebo treatment.
Assignment and masking
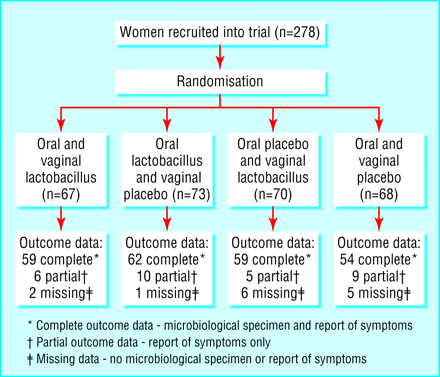
Using computer generated random numbers, we assigned women to receive one of four combinations of oral and vaginal treatment (see figure) in block randomised groups of eight. To ensure allocation concealment, an independent research fellow oversaw the labelling and packaging of trial treatments and held the randomisation schedule. Recruiters were instructed to use consecutively numbered interventions. Participants, recruiters, and microbiology staff were blinded to group assignment. A survey question assessed the success of masking. Community recruiters did not have access to microbiology results, and microbiologists did not know participants' symptom status.
Outcome measures
On recruitment to the study, participants underwent a baseline survey and provided a self collected vaginal swab. The survey and swab were repeated four days after completion of the interventions, generally 14 days after recruitment or at the time of developing symptoms of vulvovaginitis. The appropriate timing of this second swab was unclear, with no published evidence to guide the decision. The first 25 participants took additional swabs at 21 and 28 days after recruitment, but, as no cases of vulvovaginitis developed after 14 days, later participants took a follow up swab only at 14 days.
Swabs were initially examined microscopically—by making a Gram stained smear—followed by inoculation onto CHROMagar medium (CHROMagar France),10 which was incubated at 35°C for two days. We identified Candida species by the specific colour of colonies on CHROMagar or by means of ID32C kit (bioMerieux).11
The primary outcome was symptomatic vulvovaginal candidiasis, defined as symptoms (vaginal itch or irritation with or without discharge) plus candida isolated from the follow up vaginal swab. Asymptomatic women at day 14 with candida cultured from their follow up swab were contacted regarding any later symptoms but were not included as cases since a swab was not obtained if and when they were symptomatic. Any participants who did not provide a follow up survey or swab were followed up within a week and asked about vulvovaginitis symptoms.
Statistical analysis
We calculated sample size to make two comparisons—lactobacillus and placebo components in both oral and vaginal treatments. A factorial design allowed examination of two independent interventions in the same number of participants as a single intervention, as no interaction was anticipated. On the basis of the 23% incidence of post-antibiotic vulvovaginitis that we found in our earlier survey,4 we calculated that, to detect a clinically meaningful halving of infections to 11.5% for active treatment, we needed outcome data for 372 women for 80% power and significance level of 5% for a two sided test. To allow for 25% drop out, we therefore needed to recruit 496 women (124 in each of the four combinations of treatments).
Our analysis was on the basis of intention to treat, with two pre-specified comparisons—(a) oral lactobacillus versus placebo and (b) vaginal lactobacillus versus placebo. The Data Monitoring Committee reviewed our interim analyses after recruitment of 126 and 252 women. It used O'Brien and Fleming's procedure and stopping rules,12 with a nominal P value of 0.0005 at the first analysis and 0.014 at the second. It also used stochastic curtailment methods to estimate the conditional power of the study if it were completed as planned given what had been observed in each interim analysis.13 To allow for the loss of power due to three planned analyses, we inflated the sample by a factor of 1.017 from 372 to 380 women.
For our analyses, we used Stata version 7 (StataCorp, College Station, TX). To compare differences in baseline characteristics between women who did and did not provide outcome data, we used Pearson's χ2 statistic for categorical variables and two sample t tests for age.
We used logistic regression to examine the effects of the two interventions. As this was a factorial design, we adjusted the effect of each intervention for the other in the model. The factorial design also allowed us to test for interaction between the two treatment factors, but the study was not specifically powered to detect such an effect.
We performed sensitivity analyses whereby participants who could not be contacted and those who reported symptoms of vulvovaginitis but did not provide a vaginal swab at follow up were, in the first scenario, assumed not to have vulvovaginitis and, in the second scenario, assumed to have vulvovaginitis. Asymptomatic participants who did not complete the trial were assumed not to have vulvovaginitis.
Results
Viability of the lactobacilli and correct labelling of the interventions were confirmed in three independent tests. After the second interim analysis showed no evidence of effect with active treatment, the Data Monitoring Committee recommended terminating the trial early. At this stage, the committee had 62% of the expected data, and stochastic curtailment methods showed that if the trial proceeded to full enrolment the chances of detecting a significant reduction in vulvovaginitis with oral or vaginal lactobacillus treatment were less than 0.032 and 0.0006 respectively.
A total of 278 women were randomised (26 while awaiting the second interim analysis results). Because of the recruitment method used, it was not practical to document the entire eligible population. However, an audit of 132 eligible women by 13 recruiters showed that 25% agreed to randomisation and that non-participants were similar to participants in key characteristics (age, use of oestrogen based drugs, and antibiotic prescribed) (results not shown).
Complete data for the main outcomes were available for 235 women; partial data were available for 29 (report of symptoms but no vaginal swab at follow up), of whom six reported symptoms of vulvovaginitis; and no outcome data were available for 14 women. The figure shows the numbers of participants by treatment group. Table 1 shows the reasons for women's withdrawal from the trial and non-compliance with treatment. Table 2 shows participants' key baseline characteristics, which were reasonably balanced between the treatment groups.
Reasons for withdrawal from trial of oral and vaginal lactobacillus for preventing post-antibiotic vulvovaginitis among 278 women and levels of poor compliance. Values are numbers of women
Baseline characteristics of 278 women in trial of oral and vaginal lactobacillus for preventing post-antibiotic vulvovaginitis by treatment group. Values are numbers (percentages*) unless stated otherwise
Women who did not supply complete outcome data were more likely to be younger, to smoke, and not to have completed tertiary education than those who supplied full data (table 3). Among the 234 participants who gave the information, good compliance (“most” or “all” treatments taken) was reported by 225 (96%) for prescribed antibiotics, by 201 (86%) for the oral intervention, and by 184 (79%) for the vaginal intervention, with no significant difference across factorial groups. More than half the participants answered “Don't know” to the question of which treatment group they were assigned to; 64/231 (28%) correctly guessed they were in the active oral group, 46/230 (20%) correctly guessed they were in the active pessary group, and 21/228 (9%) correctly guessed their factorial group.
Comparison of baseline characteristics of women with and women without complete outcome data from trial of oral and vaginal lactobacillus for preventing post-antibiotic vulvovaginitis. Values are numbers (percentages*) unless stated otherwise
Effects of the interventions
Overall, 55/235 of the participants (23% (95% confidence interval 18% to 29%)) developed post-antibiotic vulvovaginitis. Table 4 shows the results for the factorial groups. The odds ratios for developing vulvovaginitis while taking oral lactobacillus was 1.06 (95% confidence interval 0.58 to 1.94) and while taking vaginal lactobacillus was 1.38 (0.75 to 2.54).
Number of cases of post-antibiotic vulvovaginitis in women taking oral and vaginal lactobacillus, by treatment group
Sensitivity analysis
Table 5 shows the sensitivity analyses for women without complete outcome data, assuming either all such women were not affected or that those reporting symptoms and those with unknown outcomes had vulvovaginitis. There was no evidence of an interaction between oral and vaginal treatments (results not shown). Only three of the 29 asymptomatic women with a positive follow up swab for candida reported symptoms of vaginitis after the end of the trial.
Sensitivity analyses for numbers of cases of post-antibiotic vulvovaginitis in women taking oral and vaginal lactobacillus. Participants who did not give complete outcome data were either viewed as all non-cases, or those who reported symptoms or had unknown outcomes were viewed as cases
Discussion
This study found that oral and vaginal probiotic treatments with a main constituent of Lactobacillus rhamnosus were ineffective in preventing post-antibiotic vulvovaginal candidiasis.
Strengths of study
These results should be generalisable to non-pregnant premenopausal women, as recruitment was through a wide, community based network with broad inclusion criteria (and information from a small number of eligible women who did not participate showed that they had similar key characteristics associated with the development of vulvovaginal candidiasis). Randomisation, blinding, and allocation concealment were effective, and compliance with antibiotic and intervention treatments was high. We obtained at least some outcome data from 95% of participants. Sensitivity analyses that included women with missing outcome data did not alter the significance of the results. Self collection of vaginal specimens by patients for diagnosis of infections is a convenient, validated method for many potential pathogens.14
The decision to end the trial prematurely was clear after consideration of the conditional power calculations at the second interim analysis. It was considered unethical to continue recruitment to a trial with no potential benefit.15
Limitations of study
A limitation of this trial was inadequate documentation of the entire eligible population, although audit results confirmed that participants were similar to non-participants. We might have missed some late cases of vulvovaginitis because of follow up at 14 days, but all women with a vaginal swab at follow up that was positive for candida were again questioned about symptoms of vulvovaginitis. Reliance on self obtained vaginal swabs may have led to our underestimating the number of cases, but this should have affected the active treatment and placebo arms equally.
Our results derive from one formulation and dose of lactobacillus. Arguably, it is not appropriate to generalise to other probiotics advocated to prevent vulvovaginitis. However, there is no scientifically plausible explanation to suggest how lactobacilli could be effective. The exact role of these predominant vaginal bacteria in vaginal health is not yet certain.16 17 Unlike in bacterial vaginosis, which is associated with increased vaginal pH and where hydrogen peroxide producing lactobacilli play a role in treatment,18 in acute vulvovaginal candidiasis the vaginal pH remains normal19 and lactobacilli are as likely to be the dominant bacteria as in asymptomatic women.20 Up to 20% of asymptomatic women have candida present, and the exact mechanism by which antibiotics increase the risk of developing symptomatic candidiasis is unknown.19 This uncertainty reflects lack of understanding of the complex role of immunity in vaginal candidiasis, which seems to function at local levels rather than systemically.21
Conclusions
Even if a viable hypothesis existed to explain a role for lactobacilli, to be effective, oral lactobacilli would need to survive gastric acidity and ingested antibiotics and then colonise the lower bowel and vagina. Although colonisation may occur after prolonged ingestion of yoghurt,22 this does not seem likely in the 10 day time frame of this study. Also lactobacilli exhibit host specificity and colonisation potential17: not all species can colonise the human gastrointestinal tract and vagina. Lactobacillus pessaries are also subject to the effects of antibiotics. Unless a biologically plausible theory develops, further research in this subject is unlikely to be fruitful.
The use of lactobacillus in post-antibiotic vulvovaginitis is an example of a treatment that has widespread use despite lack of a biologically plausible basis or evidence of effectiveness. Our results should prompt health professionals to inform women that lactobacillus is unlikely to prevent post-antibiotic vulvovaginitis and that they should consider using proved antifungal treatment if symptoms develop.
What is already known on this topic
Post-antibiotic vulvovaginitis is a common problem for women and may affect their concordance with prescribed antibiotics
Products containing lactobacillus species are widely used in the belief that they will prevent post-antibiotic vulvovaginitis, but there is no evidence to support this belief
What this study adds
This randomised controlled trial shows that neither oral nor vaginal forms of lactobacillus taken during and for four days after antibiotics were effective in preventing post-antibiotic vulvovaginitis
Unless a biologically plausible theory develops to explain a role for lactobacillus, further research in this subject is unlikely to be fruitful
Footnotes
-
We thank all the women, general practitioners, and pharmacists who participated in this study. Nutrition Care and Institut Rosell kindly donated the treatments and placebos used. Health Care Network developed a software prompt for use in trial recruitment. Mayne Health and Gribbles Pathology provided free transportation of some microbiological specimens. We thank Professor Judith Lumley for chairing the Data Monitoring Committee and Ms Cate Nagle for managing the randomisation schedule.
-
Contributors MP and JG initiated the study and, together with PC, SGrover, SGarland, and SH developed the study design and wrote the grant application. MP and PO'M worked on data collection and literature searching. PCand MP worked on statistical analysis. All authors contributed to data interpretation, and manuscript preparation. MP is guarantor for the study.
-
Funding The study was funded by a Quality Use of Medicine Scholarship funded by the Australian Commonwealth Department of Health and Aging; the Shepherd Foundation; the Royal Australian College of General Practitioners; Nutrition Care; Institut Rosell; Health Care Network; and Mr Sean Howard.
-
Competing interests MP received active treatments, placebos, and funding for this research from Nutrition Care and Institut Rosell.
-
Ethical approval The Royal Australian College of General Practitioners and the Royal Women's Hospital, Melbourne, granted ethical approval.